Povidone Iodine Nasal Spray and COVID-19: Studies Conclude that Povidone Iodine Might Be Helpful in Improving Outcomes
Povidone Iodine has been well documented as one of the most effective of all antimicrobials available. Hospitals and medical facilities worldwide use povidone-iodine (Betadine) as a standard of care in infection control, even though it contains very small amounts of Iodine. Betadine (PVP-1) has been in commercial use since 1955 and is on the World Health Organization’s list of essential medicines.
For more than 50
years, PVP-1 has been used as an essential medicine in hospitals and
health-care clinics as a scrub for surgeries as well as hand
disinfection for surgical personnel. It has been an integral antiseptic
as a wound disinfectant.
PVP-1 has also been shown to be highly effective in the treatment of periodontal disease. Jørgen Slots wrote
that it is a valuable antiseptic in its treatment of periodontal
disease and a variety of other oral infections. His research has shown
that it kills all periodontal pathogens in vitro within 15–20 seconds.
Slots also says that it exhibits a wide viricidal spectrum, covering
both nonenveloped and enveloped viruses, including the
periodontopathogenic cytomegalovirus.
Source:10.1111/j.1600-0757.2011.00429.x
Fortunately, there is a
new generation of recently patented iodine-based antiseptics that
overcome the negative side effects of PVP-1. A new aqueous formulation
with 100 times more I2 than contained in PVP-1 is now available, and the
nonbiocidal content has been reduced from 31,600 ppm to a few hundred
ppm. This new formulation increases its effectiveness, safety, and shelf
life, while also reducing staining, toxicity, bad taste, and potential
irritancy. It is now available in mouthrinses, concentrates, nasal
sprays, and hand sanitizers offered by IoTech International.
Source: IoTech International, Boca Raton, Florida
 |
| Buy on Amazon |
Source:10.1002/jps.3030440315
In a study by the Institute for Antiviral Research at Utah State
University, it was demonstrated that the COVID-19 virus can be
completely inactivated with a 30-second exposure to a 100 ppm molecular
iodine (I2) mouthrinse.1 This
in vitro study opens the door for further research and the need to
review past and current research about the efficacy of I2 as an antimicrobial therapeutic agent. Molecular iodine is the only species of iodine that is antimicrobial.
Source: https://pubchem.ncbi.nlm.nih.gov/patent/US2018360048
 |
| Buy on Amazon |
Research has continued to show iodine to be effective against viruses. In 1943, Dunham and MacNeal demonstrated that vaccinia virus could be killed with a 3% tincture of iodine. In 1955, Louis Gershenfeld found that tincture of iodine was the most the effective antiseptic found to quickly destroy the poliovirus. He reported that he developed a mouthwash capable of killing the virus using concentrations of free iodine. These landmark studies and others demonstrate iodine’s efficacy as an antiviral.9
Source: Iodine: the forgotten weapon against influenza viruses
Recently, much research has been
published on the effects PVP-1 has on the COVID-19 virus and its rapid
inactivation upon exposure. Multiple studies have shown that
povidone-iodine with its small amount of I2 has quick and effective
antiviral qualities and is sufficient to be used as a proactive
preventive therapy against COVID-19. It has many uses including hand and
skin sanitizing, oral rinsing, throat spraying, gargling, and nasal
spraying to inactivate the virus.
Sources:
Currently, the most common source of iodine disinfection is PVP-1, but there are limitations of use due to its staining, toxicity, and taste. This new generation of iodine products is now available and has eliminated these negative effects by isolating, stabilizing, and concentrating the bioactive I2. This has enormous implications in the management and treatment of many microbial-sourced diseases. Since I2 has been proven to completely inactivate the COVID-19 virus, it seems prudent that it be used in medical and dental environments.
Source:10.1016/j.mehy.2020.109860
Most doctors know the need to focus now on early treatment as the most immediate and practical way to reduce hospitalizations and death. However, some doctors and media channels argue that there is very little evidence to support the use of povidone-iodine to prevent or treat COVID-19.Below, we look at the studies for Povidone-Iodine (PVP) and COVID-19:
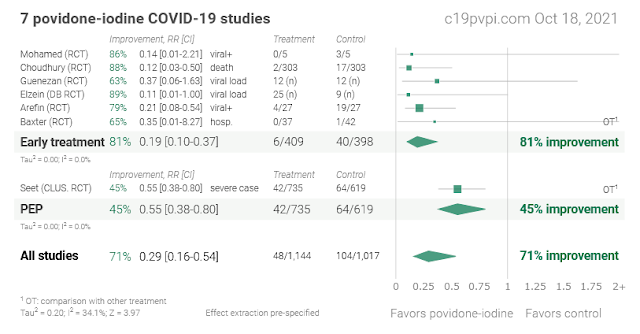
8 Povidone Iodine against COVID-19 Studies
1. Rapid initiation of nasal saline irrigation: hospitalizations in COVID-19 patients randomized to alkalinization or povidone-iodine compared to a national dataset
Baxter et al., medRxiv, doi:10.1101/2021.08.16.21262044 (Preprint)
Small RCT 79 PCR+ patients 55+ comparing pressure-based nasal irrigation with povidone-iodine and sodium bicarbonate, showing improved recovery with povidone-iodine, and 0/37 COVID-19 related hospitalizations for povidone-iodine compared to 1/42 for sodium bicarbonate. NCT04559035.
2. Virucidal effect of povidone iodine on COVID-19 in the nasopharynx: an open-label randomized clinical trial
Arefin et al., Indian Journal of Otolaryngology and Head & Neck Surgery, doi:10.1007/s12070-021-02616-7 (Peer Reviewed)
RCT with 189 patients showing significantly greater viral clearance with a single application of PVP-I. Authors recommend using PVP-I prophylactically in the nasopharynx and oropharynx. NCT04549376
3. Positive impact of oral hydroxychloroquine and povidone-iodine throat spray for COVID-19 prophylaxis: an open-label randomized trial
Prophylaxis RCT in Singapore with 3,037 low risk patients, showing lower serious cases, lower symptomatic cases, and lower confirmed cases of COVID-19 with all treatments (ivermectin, HCQ, PVP-I, and Zinc + vitamin C) compared to vitamin C.Meta-analysis of vitamin C in 6 previous trials shows a benefit of 16%, so the actual benefit of ivermectin, HCQ, and PVP-I may be higher. Cluster RCT with 40 clusters.There were no hospitalizations and no deaths. NCT04446104.
4. In vivo evaluation of the virucidal efficacy of chlorhexidine and povidone-iodine mouthwashes against salivary SARS-CoV-2. A randomized-controlled clinical trial
Small RCT comparing mouthwashing with PVP-I, Chlorhexidine, and water, showing significant efficacy for both PVP-I and Chlorhexidine, with PVP-I increasing Ct by a mean of 4.45 (p < 0.0001) and Chlorhexidine by a mean of 5.69 (p < 0.0001), compared to no significant difference for water.

5. Can povidone Iodine gargle/mouthrinse inactivate SARS-CoV-2 and decrease the risk of nosocomial and community transmission during the COVID-19 pandemic? An evidence-based update

6. Povidone Iodine Mouthwash, Gargle, and Nasal Spray to Reduce Nasopharyngeal Viral Load in Patients With COVID-19: A Randomized Clinical Trial
RCT of PCR+ patients with Ct<=20 with 12 treatment and 12 control patients, concluding that nasopharyngeal decolonization may reduce the carriage of infectious SARS-CoV-2 in adults with mild to moderate COVID-19. All patients but 1 had negative viral titer by day 3 (group not specified). There was no significant difference in viral RNA quantification over time. The mean relative difference in viral titers between baseline and day 1 was 75% [43%-95%] in the intervention group and 32% [10%-65%] in the control group. Thyroid dysfunction occurred in 42% of treated patients, with spontaneous resolution after the end of treatment. Patients in the treatment group were younger.

7. Effect of 1% Povidone Iodine Mouthwash/Gargle, Nasal and Eye Drop in COVID-19 patients
RCT 606 patients in Bangladesh for povidone iodine mouthwash/gargle, nasal drops and eye drops showing significantly lower death, hospitalization, and PCR+ at day 7.

8. Early viral clearance among COVID-19 patients when gargling with povidone-iodine and essential oils: a pilot clinical trial
Early viral clearance among COVID-19 patients when gargling with povidone-iodine and essential oils: a pilot clinical trial Tiny RCT with 5 PVP-I patients, gargling 30 seconds, 3x per day, and 5 control patients (essential oils and tap water were also tested), showing improved viral clearance with PVP-I.








.png)
Comments
Post a Comment