COVID antibody drugs have saved lives — so why aren’t they more popular?
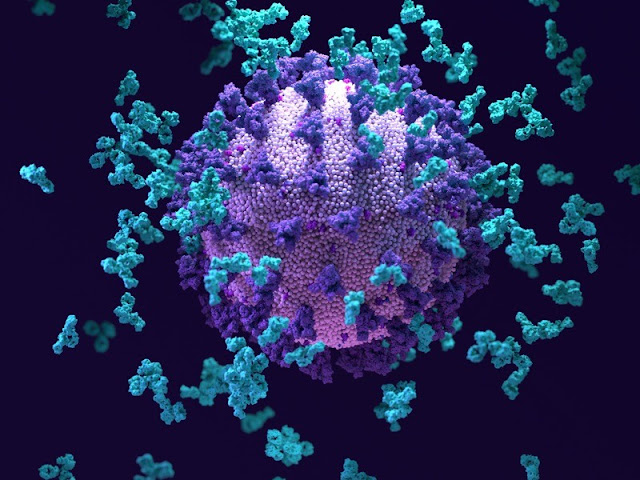
Arturo Casadevall watched aghast as the number of COVID-19 cases started to climb at the start of the pandemic. But he also saw scope for a solution. For decades, Casadevall, an infectious-disease researcher at Johns Hopkins Bloomberg School of Public Health in Baltimore, Maryland, has been working in the laboratory and in the clinic to unleash the potential of monoclonal antibody therapies — extremely precise drugs made up of the proteins that defend the body against invading microorganisms. With a new virus running rampant and no treatment options available, Casadevall hoped that antibodies would have their chance to shine.
|
| Credit: Design Cells/SPL |
The drugs rose to the occasion. By early November 2020, the US Food and Drug Administration (FDA) had issued emergency-use authorization for the first antibody to treat COVID-19, which reached patients before any vaccines or other tailored antivirals. More followed, helping to save the lives of people with COVID-19 and even staving off infection in healthy recipients. When Casadevall’s cousin came down with COVID-19 last August, Casadevall helped to lobby his cousin’s doctor to prescribe an antibody. “Antibodies need to be celebrated,” he says.
- Effectiveness of Casirivimab-Imdevimab and Sotrovimab During a SARS-CoV-2 Delta Variant Surge - A Cohort Study and Randomized Comparative Effectiveness Trial (JAMA, July 2022)
- Patients treated with monoclonal antibodies during COVID-19 delta surge had low rates of severe disease, study finds (Mayo Clinic, June 2022)
But despite the early successes, the party hasn’t started. Instead, governments and drug developers threw their weight behind vaccines, which are cheaper to make and easier to dispense; antibody drugs for COVID-19 can cost thousands of dollars a dose, compared with just a few dollars for vaccines. Globally, supply and demand for antibodies was low, and the drugs were sidelined. Even in the United States, where they’ve been used the most, they can be hard to get hold of. And as the virus started to evolve, the efficacy of the earliest antibodies waned.
Antibodies that treat cancer and immune dysfunctions are a booming, multibillion-dollar industry. But little new funding has been directed to those aimed at viruses and bacteria, and the number of infectious-disease antibodies in clinical development has flat-lined. That could be set to change: non-profit groups are hoping to support the development of antibodies as a way to prepare for future outbreaks.
Vaccines might be the ideal way to tackle a global pandemic — but they shouldn’t be the only one, says Angela Rasmussen, a virologist at the Vaccine and Infectious Disease Organization at the University of Saskatchewan in Saskatoon, Canada. “It is really crucially important that we don’t pick one horse and bet on it. We need to bet on the entire field,” she says. Antibodies have some advantages over vaccines, such as providing lasting protection in people with weakened immune systems, she says.
Antibodies will be a key solution for the next pandemic, adds Julie Gerberding, chief executive of the Foundation for the National Institutes of Health (FNIH) in Bethesda, Maryland, and former director of the US Centers for Disease Control and Prevention. “The idea of using antibodies to ward off new infectious diseases is just — to me — common sense.”
Pandemic potential
Antibodies are a pillar of the immune system. When the body encounters a viral or bacterial invader, it custom-makes these Y-shaped proteins to bind to unique markers on the invader’s surface. The two arms of the Y lock on to the offender, and the stem fires up the immune system to call for back up.
Researchers worked out how to produce monoclonal antibodies en masse some 50 years ago, by cloning the cells that make them. Ever since, drug developers have been turning them into therapies, disarming human proteins involved in conditions such as autoimmune disorders, cancer, heart disease and migraines. Adalimumab, long the world’s top-selling drug, soothes rheumatoid arthritis and other autoimmune conditions by mopping up the inflammatory protein TNF-α. Pembrolizumab, on track to overtake adalimumab’s sales, binds to a protein on the immune system’s T cells to unleash the body’s defences on cancers. Last year, the FDA approved its 100th monoclonal antibody, and these drugs collectively reap around US$150 billion in sales worldwide every year.
But despite the natural role of antibodies in deflecting pathogens, they have had few successes against infectious diseases. This is partly because specialists have prioritized the hunt for broad-spectrum drugs that can take on multiple pathogens at a time, and antibodies are suited for only a single adversary. But industry has also been deprioritizing research into infectious diseases for decades, owing to the hurdles of making money in this space — in particular, the availability of cheap generic drugs, the need to ration medicines to slow the rise of resistance and the lower purchasing power of the countries that could benefit the most.
Just a handful of the antibodies approved by the FDA target infections, including those caused by Ebola virus, respiratory syncytial virus (RSV) and the bacteria Clostridium difficile and Bacillus anthrax.
COVID-19 has put infectious-disease antibodies back in the spotlight. A front-runner was REGEN-COV — a treatment made by Regeneron Pharmaceuticals in Tarrytown, New York. The FDA authorized it for use in emergencies in late November 2020 — a month ahead of the mRNA vaccines and more than a year before bespoke antivirals made from small molecules, such as Paxlovid (a combination of nirmatrelvir and ritonavir). “The monoclonal antibody approaches were light-speed fast,” says Ann Eakin, a senior scientific officer at the US National Institute of Allergy and Infectious Diseases (NIAID) in Bethesda.
REGEN-COV comprises two antibodies — casirivimab and imdevimab — that bind to the spike protein on the surface of the SARS-CoV-2 virus, preventing it from
sneaking into host cells. In adults with mild-to-moderate COVID-19 and a high
risk of severe disease, the cocktail lowers the relative risk of
hospitalization or death by more than 70%: the rate was 1.3% in people who
received the drug, compared with 4.6% among those who did not1.
Antibody drugs can also prevent infection, both in people who have recently been exposed to the SARS-CoV-2 virus and in those who don’t respond well to vaccines. The antibody cocktail Evusheld (tixagevimab and cilgavimab), developed by AstraZeneca in Cambridge, UK, showcases these drugs’ vaccine-like capabilities: it reduces the relative risk of developing COVID-19 by more than 75% in elderly people and those with compromised immune systems, who often do not respond well to vaccines. A single dose provides at least 6 months of protection2, and possibly up to 12. Last December, Evusheld became the first antibody therapy to secure FDA authorization for pre-exposure prevention of COVID-19 in people with compromised immune systems. Evusheld is also effective as a treatment3, although it has not been authorized for that use.
Of all the COVID-19 antibodies, REGEN-COV has been the most widely used: the United States has administered around two million doses. It is also the most profitable, with sales of US$5.8 billion in the United States and a further $1.7 billion in the rest of the world (where it is sold by the Swiss drug firm Roche as Ronapreve). “Before the pandemic, most doctors and patients did not know a lot about therapeutic antibodies in infectious diseases,” says Regeneron’s senior vice-president, Christos Kyratsous. “We built a lot of awareness.”
But globally, availability is patchy. The United Kingdom has administered just 33,000 doses. Access to the drugs in middle- and low-income countries is almost non-existent, according to disclosed antibody purchases.
Resistance levels
Antibodies also have an Achilles heel. Because they are picky about their targets, they are easily out-manoeuvred by rapidly evolving viruses. “Pathogens change a couple of amino acids and the antibodies no longer bind,” says Casadevall.
The first antibody to receive FDA authorization — bamlanivimab, made by Lilly in Indianapolis, Indiana — was outflanked by the virus in five months. The REGEN-COV cocktail fared better, and was used in the United States for around 14 months, helping to fill the gap in treatment options there until antivirals arrived. But its efficacy faltered with Omicron and its use was restricted. The Evusheld combination remains effective against current variants.
For James Crowe, a viral immunologist at Vanderbilt University in Nashville, Tennessee, and discoverer of the antibodies in Evusheld, such data show that two-antibody cocktails that restrict a pathogen’s escape routes are the best way forward. As researchers become better mixologists, their concoctions will last even longer against both COVID-19 and other infectious diseases, he says.
Others contend that single antibodies — if they are designed to hit the target at the right spot — might yet win out.
When COVID-19 struck, Regeneron, AstraZeneca and other antibody developers prioritized the candidates that packed the biggest punch, with the strongest ability to neutralize the virus. Researchers then combined the best performers to make the final product. But others went for staying power, focusing on regions of the spike protein that mutate more slowly. These candidates might be less potent in animal models, but they stand a better chance of fighting off future variants of SARS-CoV-2 and perhaps even related coronaviruses, says Herbert Virgin, chief scientific officer at Vir Biotechnology in San Francisco, California. His team used this approach to discover the antibody sotrovimab4, and partnered with the UK drug firm GlaxoSmithKline in London to develop it; another partnership with a similar goal, between Lilly and AbCellera in Vancouver, Canada, yielded bebtelovimab.
Bebtelovimab is now the only antibody recommended as a treatment in the United States, and only when antivirals are unavailable. Sotrovimab is the only one in use in the United Kingdom.
These ‘broadly neutralizing’ antibodies are the future, argues Virgin. “If we had a few of those on the shelf, we might not have to isolate new antibodies when the next pandemic arises,” he says.
The idea is not new; drug developers have been hunting for such antibodies for other viruses for decades. They have tested at least a dozen candidates against HIV in clinical trials with little success, although one study showed that a two-antibody cocktail could suppress HIV levels in a subset of people5. Lasting efficacy for antibodies against SARS-CoV-2 could buoy hopes of victory against other viruses, too.
Virgin hopes that research on COVID-19 antibodies will translate into better drugs for other diseases. Vir is now tweaking sotrovimab to supercharge its ability to put the immune system into attack mode. This kind of strategy could make antibodies for other disorders such as cancer more potent — but risks sending the immune system into overdrive. If Vir can prove the approach is safe with viral targets such as SARS-CoV-2, drug developers might be tempted to use similar strategies to set the immune system on cancer cells.
Policy problems
Despite the clinical potential of infectious-disease antibodies, the pandemic exposed the difficulty of getting them to the people who need them. “There are the scientific challenges, and then there are the policy challenges. I’m glad I work on the former,” says Mark Esser, vice-president of microbial sciences at AstraZeneca.
Health-care systems have struggled to distribute COVID-19 antibodies effectively and equitably, even more so than they did with vaccines and antiviral medicines such as Paxlovid. Not only do these drugs need to be given early in the course of infection for best effect, but the first COVID-19 antibodies were also best delivered by intravenous drip. This created diagnostic, infrastructural, staffing and other bottlenecks.
Antibodies also tend to cost more than do antivirals and vaccines — around $2,100 per dose of REGEN-COV, for example, versus up to $530 for Paxlovid or $20 for the mRNA vaccine Comirnaty, which is made by Pfizer, in New York City, and BioNTech in Mainz, Germany.
But more broadly, infectious diseases have long been a losing ticket for the pharmaceutical industry. One of the problems is that no health system or pharmaceutical company wants to spend money on drugs that are used infrequently, only as last resorts. COVID-19 has provided an unprecedented windfall in terms of infectious diseases for some, but it remains hard to build a company on once-in-a-generation pandemics.
As business returns to normal, companies will keep prioritizing the most profitable drug-development opportunities. A few viruses could fit the bill. Even before the pandemic, Vir was testing antibody drugs to treat influenza and hepatitis B virus, which infects the liver. Gilead Sciences in Foster City, California, is developing the two-antibody cocktail for HIV that has shown some promise4. And AstraZeneca hopes to soon secure approval for a long-acting antibody called nirsevimab6, to protect newborns against RSV infection.
There are opportunities for infectious-disease antibodies in settings where “vaccines won’t work or won’t work well”, says Esser.
The task of preparing a drug cabinet to be ready for future pandemics is likely to fall to governments and charities, adds Crowe. To this end, he has founded the AHEAD 100 initiative, a non-profit collaboration to develop and stockpile 100 monoclonal antibodies that can protect against 25 high-risk virus families and hopefully quell would-be pandemics. He puts the price tag for this work at $2.5 billion.
The Coalition for Epidemic Preparedness Innovations (CEPI) in Oslo, another non-profit group that is investing billions of dollars into vaccines for pandemics, has also added antibodies to its remit. It could soon start funding work on antibodies against four priority pathogens, which are yet to be identified. “We are at a major historical tipping point, in which antibodies are going to become one of the principal tools that we use to manage infectious diseases,” says Crowe.
Eakin expects that it is just a matter of time before infectious-disease antibodies get more financial support. So far, public and private funders have prioritized vaccine platforms; in May, NIAID invested $577 million in small-molecule antivirals, but only because they were so much slower to progress through the pipeline than were vaccines and antibodies. Antibodies are for now stuck in the neglected middle, but Eakin doesn’t think they will stay there.
Gerberding, too, hopes that more funding is coming for infectious-disease antibodies — as well as for other pillars of pandemic preparedness. “We’re just scratching the surface of what we have the capability of doing, but we don’t want to pay the bill. If we haven’t learnt yet from COVID that paying the bill would have been worth it, I don’t know what it’s going to take.”
Nature 606, 854-856 (2022)
doi: https://doi.org/10.1038/d41586-022-01735-7
References
PubMed Article Google Scholar
Levin, M. J. et al. N. Engl. J. Med. 386, 2188–2200 (2022).
PubMed Article Google Scholar
Montgomery, H. et al. Lancet Resp. Med. https://doi.org/10.1016/S2213-2600(22)00180-1 (2022).
Article Google Scholar
Pinto, D. et al. Nature 583, 290–295 (2020).
PubMed Article Google Scholar
Sneller, M. C. et al. Nature 606, 375–381 (2022).
PubMed Article Google Scholar
Hammitt, L. L. et al. N. Engl. J. Med. 386, 837–846 (2022).
PubMed Article Google Scholar







.png)

Comments
Post a Comment