Treatment of Omicron Variant (October 2022)
As of the week ending October 8, 2022, BA.5 represent an estimated 79.2% of the SARS-CoV-2 variants currently circulating in the United States, according to the CDC. The BA.4.6 Omicron subvariant is the second most prevalent with 13.6% of cases originating from the pathogen.
Data suggest that these lineages could be more transmissible than previous Omicron sublineages. However, there is no evidence currently available to suggest that BA.4 and BA.5 cause more severe disease than other variants or Omicron lineages. US CDC is continuing to assess the impact that BA.4 and BA.5 have on public health.
BA.5 is one of many Covid-19 Omicron subvariants to emerge since last winter. The subvariant is also driving up cases in parts of Europe and North America and has become the dominant U.S. Omicron strain. This version of the virus is believed to spread particularly easily, fueled in part by its ability to evade immunity built up from vaccines and prior infections.
The research found that the antibodies were several times less effective against the new sub-variants than they were against the original Omicron strain. However, antibodies produced by the 15 individuals who had been vaccinated against COVID-19 were more effective than those from people who had recovered from natural infection alone.
COVID-19 Omicron BA4 and BA5 Variant Symptoms
According to the US CDC (updated August 2022), people with COVID-19 have had a wide range of symptoms reported – ranging from mild symptoms to severe illness. Symptoms may appear 2-14 days after exposure to the virus. Anyone can have mild to severe symptoms.Possible symptoms include:
- Fever or chills
- Cough
- Shortness of breath or difficulty breathing
- Fatigue
- Muscle or body aches
- Headache
- New loss of taste or smell
- Sore throat
- Congestion or runny nose
- Nausea or vomiting
- Diarrhea
Related: Flu vs COVID-19
Take a Test
Between 48 and 72 hours after potential COVID exposure or at the first sign of any symptoms, people should take either a rapid antigen or PCR test. “There should be a very low threshold for testing yourself to see whether you have COVID or whether you have something else,” says Amesh Adalja, a senior scholar at the Johns Hopkins Center for Health Security. COVID can look very like a cold, influenza or allergies, but the treatments for each are different.
PCR tests are more sensitive but harder to get, and taking a rapid antigen test at home usually suffices, the experts say. If the first test is negative, people should wait two days (behaving cautiously in the meantime) and take another one as Myers did. If it is COVID, the viral load will increase in that time. “Nothing in life is perfect, nor are the rapid antigen tests, but they’re pretty darn good at picking up contagious levels of virus,” says Lucy McBride, a practicing primary care doctor in Washington, D.C. (Lists of free test locations are available on the Test to Treat locator Web site provided by the U.S. Department of Health and Human Services.)
When people test positive, it is a good idea for them to call a primary care physician if they have one. Doctors can provide guidance on treatment and update medical histories. They will also report the result to public health authorities so that it is included in case counts. At a minimum, people should track the date that symptoms began and the date of a positive test.
Over-the-Counter Help
Most people who get COVID will be just fine at home. “If you’re vaccinated and boosted and generally healthy, people do very well,” McBride says. Over-the-counter medications will not treat COVID directly but can help manage symptoms. Doctors recommend acetaminophen (Tylenol) or nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen (Advil or Motrin) to bring down fevers and relieve achiness. Early in the pandemic, there were reports that ibuprofen made COVID worse, but those have not been substantiated. NSAIDs are only intended to be taken for short periods, however, because they have more side effects than acetaminophen, and they are not safe for everyone. People who take other medications should consult with a doctor before taking NSAIDs. Antihistamines or cold medications such as DayQuil can be used to relieve congestion and cough.
Getting enough rest is critical, as is drinking enough fluids, which prevent dehydration and reduce cough.
Having a pulse oximeter at home would also be useful to calculate the amount of oxygen in a person’s blood without having to draw a blood sample. If oxygen levels fall below 95 per cent, that would be a sign to visit the hospital.
Thermometers, tissues and hand sanitizer are other supplies often recommended to keep at home while experiencing a COVID-19 infection. Finally, it won’t hurt to take vitamin supplements as deficiencies might be associated with the severe form of COVID-19.
Using Prescription Drugs
One of the most significant recent changes in the COVID landscape is the wider availability of authorized outpatient treatments. The antiviral drug Paxlovid reduced risk of hospitalization and death by 89 percent in a clinical trial, and it is a pill, which makes it relatively easy to take. The drug cannot be taken with many common medications such as statins or blood thinners, however.
Monoclonal antibodies also significantly reduce the risk of severe disease, but they require an injection or infusion. And not every patient is eligible to get these treatments. Both have been authorized for people who are at higher risk of severe illness because of age (65 and older) or underlying health conditions such as high blood pressure or lung disease. To date, there is no evidence that these treatments benefit people who are young or otherwise healthy. Either treatment is available by prescription or at Test to Treat sites nationwide for people who are eligible.
Older version of monoclonal antibodies, such as Regeneron’s treatment (Imdevimab + Casirivimab), could fail or partially fail because they target parts of the virus that will have mutated.
Based on a lab study (NEJM, July 2022) using the live-virus Focus Reduction Neutralization Testing (FRNT) method, bebtelovimab seems to be the most promising monoclonal antibody against the BA 5 subvariant.
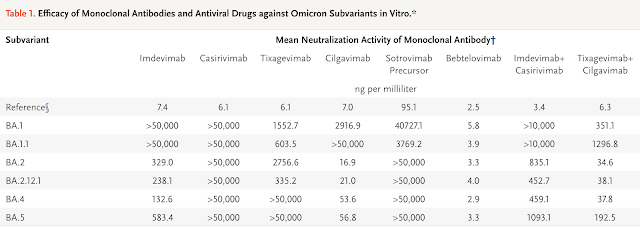
Overall, the study data also suggest that the three antiviral drugs remdesivir, molnupiravir, and nirmatrelvir may still have therapeutic value against the sublineages BA.2.12.1, BA.4, and BA.5 of SARS-CoV-2 omicron variants.
Do take note of the limitation of this study as this is a non-clinical study (not in humans). There is lack of clinical data on the efficacy of these monoclonal antibodies and antiviral drugs for the treatment of patients infected with BA.4 or BA.5 subvariants. Therefore, the selection of monoclonal antibodies or anti-virals to treat patients who are infected should be carefully considered based on the potential risks as compared to its potential benefits.
To be effective, these medications must be started soon after symptoms begin (within five days and seven days, respectively). “[People who might be eligible] should call their provider right away,” says Raymund Razonable, an infectious disease specialist at the Mayo Clinic. “These treatments work best when the patients are not feeling so bad.” Side effects of Paxlovid are uncommon but include diarrhea and a metallic taste in the mouth.
Related: Paxlovid vs Bebtelovimab
Other At-Home Treatments
You can find a listing of doctors who can prescribe necessary home isolation medications on Find a Provider post.
There are many COVID-19 treatment protocols out there on the internet. We have reviewed many protocols and believe the FLCCC I-CARE protocol is the easiest and effective to follow.
Always consult your trusted medical professional before you take any medication or supplement.
Emergency warning signs
Individuals are reminded to seek immediate medical attention (go to a hospital’s emergency department) if they develop emergency warning signs of COVID-19 such as:- Difficulty breathing
- Persistent pain or pressure in the chest
- Bluish lips or face
- New confusion or inability to arouse
Key Takeaway
As soon as you have symptoms, consult your healthcare provider and start treatment as early as possible. If treatment is delayed i.e. after 5 days of symptoms, your chances of severe COVID are higher.
Take a Test
Between 48 and 72 hours after potential COVID exposure or at the first sign of any symptoms, people should take either a rapid antigen or PCR test. “There should be a very low threshold for testing yourself to see whether you have COVID or whether you have something else,” says Amesh Adalja, a senior scholar at the Johns Hopkins Center for Health Security. COVID can look very like a cold, influenza or allergies, but the treatments for each are different.PCR tests are more sensitive but harder to get, and taking a rapid antigen test at home usually suffices, the experts say. If the first test is negative, people should wait two days (behaving cautiously in the meantime) and take another one as Myers did. If it is COVID, the viral load will increase in that time. “Nothing in life is perfect, nor are the rapid antigen tests, but they’re pretty darn good at picking up contagious levels of virus,” says Lucy McBride, a practicing primary care doctor in Washington, D.C. (Lists of free test locations are available on the Test to Treat locator Web site provided by the U.S. Department of Health and Human Services.)
When people test positive, it is a good idea for them to call a primary care physician if they have one. Doctors can provide guidance on treatment and update medical histories. They will also report the result to public health authorities so that it is included in case counts. At a minimum, people should track the date that symptoms began and the date of a positive test.
Over-the-Counter Help
Most people who get COVID will be just fine at home. “If you’re vaccinated and boosted and generally healthy, people do very well,” McBride says. Over-the-counter medications will not treat COVID directly but can help manage symptoms. Doctors recommend acetaminophen (Tylenol) or nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen (Advil or Motrin) to bring down fevers and relieve achiness. Early in the pandemic, there were reports that ibuprofen made COVID worse, but those have not been substantiated. NSAIDs are only intended to be taken for short periods, however, because they have more side effects than acetaminophen, and they are not safe for everyone. People who take other medications should consult with a doctor before taking NSAIDs. Antihistamines or cold medications such as DayQuil can be used to relieve congestion and cough.Getting enough rest is critical, as is drinking enough fluids, which prevent dehydration and reduce cough.
Having a pulse oximeter at home would also be useful to calculate the amount of oxygen in a person’s blood without having to draw a blood sample. If oxygen levels fall below 95 per cent, that would be a sign to visit the hospital.
Thermometers, tissues and hand sanitizer are other supplies often recommended to keep at home while experiencing a COVID-19 infection. Finally, it won’t hurt to take vitamin supplements as deficiencies might be associated with the severe form of COVID-19.
Using Prescription Drugs
One of the most significant recent changes in the COVID landscape is the wider availability of authorized outpatient treatments. The antiviral drug Paxlovid reduced risk of hospitalization and death by 89 percent in a clinical trial, and it is a pill, which makes it relatively easy to take. The drug cannot be taken with many common medications such as statins or blood thinners, however.Monoclonal antibodies also significantly reduce the risk of severe disease, but they require an injection or infusion. And not every patient is eligible to get these treatments. Both have been authorized for people who are at higher risk of severe illness because of age (65 and older) or underlying health conditions such as high blood pressure or lung disease. To date, there is no evidence that these treatments benefit people who are young or otherwise healthy. Either treatment is available by prescription or at Test to Treat sites nationwide for people who are eligible.
Older version of monoclonal antibodies, such as Regeneron’s treatment (Imdevimab + Casirivimab), could fail or partially fail because they target parts of the virus that will have mutated.

Overall, the study data also suggest that the three antiviral drugs remdesivir, molnupiravir, and nirmatrelvir may still have therapeutic value against the sublineages BA.2.12.1, BA.4, and BA.5 of SARS-CoV-2 omicron variants.
Do take note of the limitation of this study as this is a non-clinical study (not in humans). There is lack of clinical data on the efficacy of these monoclonal antibodies and antiviral drugs for the treatment of patients infected with BA.4 or BA.5 subvariants. Therefore, the selection of monoclonal antibodies or anti-virals to treat patients who are infected should be carefully considered based on the potential risks as compared to its potential benefits.
To be effective, these medications must be started soon after symptoms begin (within five days and seven days, respectively). “[People who might be eligible] should call their provider right away,” says Raymund Razonable, an infectious disease specialist at the Mayo Clinic. “These treatments work best when the patients are not feeling so bad.” Side effects of Paxlovid are uncommon but include diarrhea and a metallic taste in the mouth.
Related: Paxlovid vs Bebtelovimab
Other At-Home Treatments
You can find a listing of doctors who can prescribe necessary home isolation medications on Find a Provider post.
There are many COVID-19 treatment protocols out there on the internet. We have reviewed many protocols and believe the FLCCC I-CARE protocol is the easiest and effective to follow.
Always consult your trusted medical professional before you take any medication or supplement.
Emergency warning signs
- Difficulty breathing
- Persistent pain or pressure in the chest
- Bluish lips or face
- New confusion or inability to arouse
Key Takeaway
As soon as you have symptoms, consult your healthcare provider and start treatment as early as possible. If treatment is delayed i.e. after 5 days of symptoms, your chances of severe COVID are higher.







.png)

Comments
Post a Comment